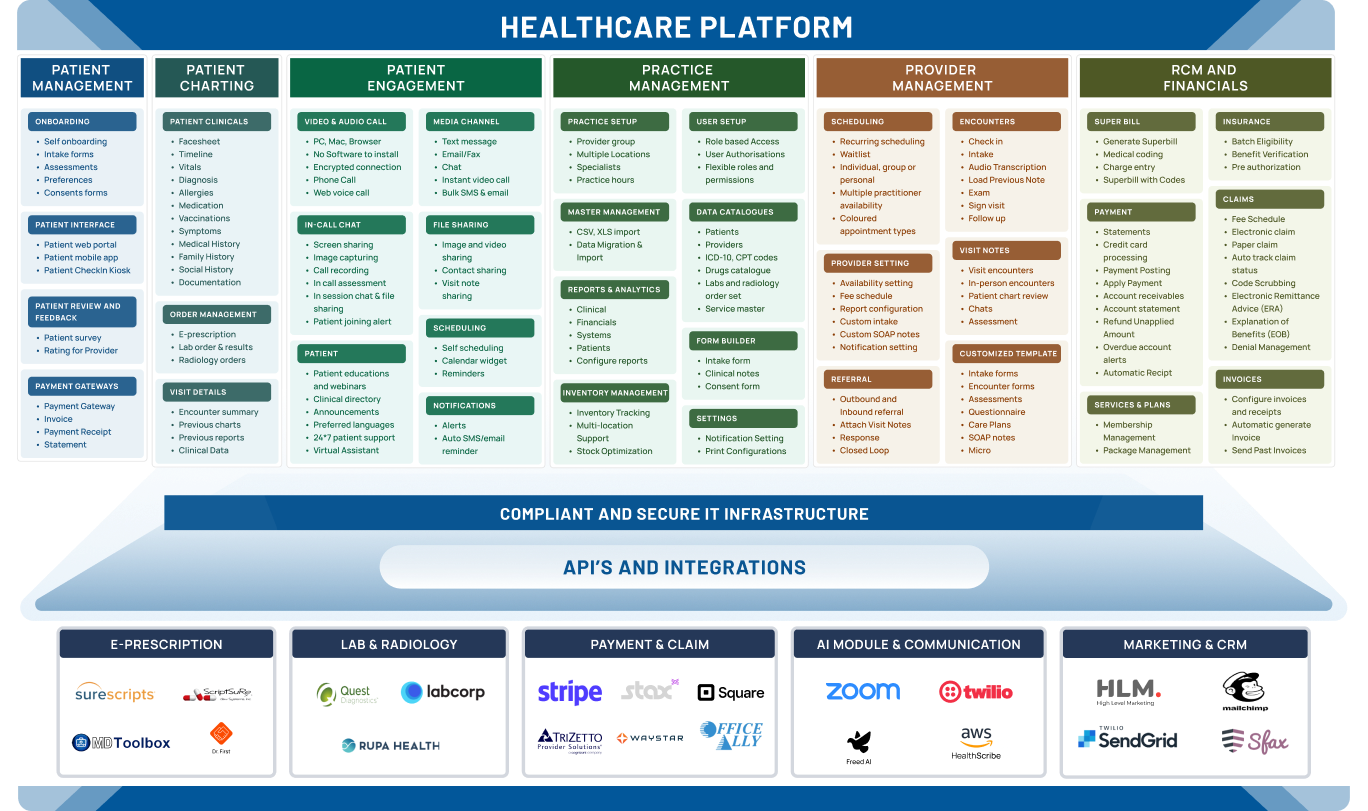
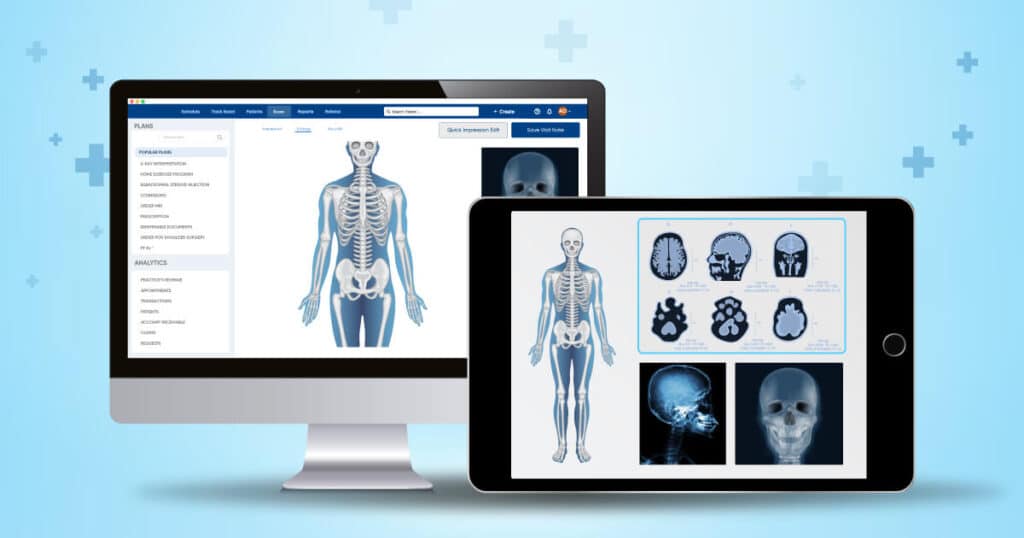
Build a secure and compliant orthopedic EHR system that meets industry standards while protecting patient data and ensuring reliable workflows.
HIPAA Compliance
Protect patient health information by adhering to strict privacy and security safeguards.
- Civil penalties up to $1.5M annually
- Risk of data breaches and lawsuits
- Damage to provider reputation and trust
- Potential loss of business licenses
- Increased malpractice insurance premiums
- Safeguards patient privacy and data security
- Ensures legal and regulatory compliance
- Builds provider and patient trust
- Enables secure system interoperability
- Positions you for future regulations
- Encrypt PHI at rest and in transit
- Role-based access control implementation
- Audit logs for all system interactions
- Regular HIPAA compliance audits and checks
- Staff training on HIPAA best practices
Our HIPAA Compliance Partners
ONC Certification
Ensures that your healthcare software meets federal standards for safety, usability, and interoperability.
- Ineligibility for federal healthcare programs
- Loss of credibility among providers
- Limited adoption by hospitals and practices
- Risk of software rejection in the U.S. market
- Competitive disadvantage against certified vendors
- Validates usability, safety, and interoperability
- Enables providers to qualify for incentives
- Ensures EHR/healthcare systems meet federal standards
- Builds market acceptance and trust
- Enhances nationwide interoperability initiatives
- Develop per ONC-ACB certification criteria
- Conduct usability and safety testing
- Ensure integration with certified APIs
- Align with ONC interoperability requirements
- Maintain certification through updates and audits
FHIR/HL7
The modern backbone of real-time, interoperable healthcare data exchange.
- Inability to exchange clinical data
- Regulatory compliance failures
- Patient care delays due to poor data access
- Loss of partnerships with health systems
- Missed opportunities in health app integrations
- Enables real-time healthcare data exchange
- Standardizes communication across EHRs and apps
- Enhances care coordination and interoperability
- Supports patient-centric, connected care ecosystem
- Facilitates integration with health apps and wearables
- Implement FHIR APIs for modern apps
- Enable HL7 v2/v3 messaging protocols
- Ensure compatibility with third-party systems
- Test interoperability across multiple vendors
- Update systems for evolving HL7 standards
Our FHIR-HL7 Compliance Partners
21st Century Cures Act
Empowers patients with data access and drives interoperability across systems.
- Penalties for information blocking ($1M per violation)
- Legal action by regulators or patients
- Loss of provider partnerships
- Increased compliance monitoring costs
- Negative press for non-transparent practices
- Gives patients secure access to health records
- Promotes data sharing without barriers
- Prevents anti-competitive practices in healthcare IT
- Improves transparency in patient care
- Encourages innovation through open access
- Enable patient-facing APIs and portals
- Ensure no artificial data blocking practices
- Maintain open data access policies
- Regularly review compliance with Cures Act rules
- Integrate audit logs to prove compliance
Our 21st Century Cures Act Compliance Partners
Medical Coding Standards
Standardizes clinical language to ensure accurate billing, reporting, and care.
- Incorrect billing and revenue losses
- Claim denials and payment delays
- Legal risks from fraudulent coding errors
- Loss of payer/provider trust
- Regulatory audits leading to penalties
- Ensures accurate clinical documentation
- Standardizes reporting for billing/reimbursement
- Facilitates research and analytics accuracy
- Enables smooth payer-provider coordination
- Supports population health and outcomes research
- Integrate ICD-10, CPT, SNOMED, LOINC codes
- Automate coding with validation tools
- Train staff for coding compliance
- Update codes with regulatory changes
- Perform audits to catch coding errors
Our Medical Coding Standards Adherence Partners
SOC 2 Type II
Validates your system’s operational security, availability, and data integrity controls.
- Loss of enterprise and B2B contracts
- Increased data breach risks
- Failure to meet client audit requirements
- Damaged reputation with healthcare clients
- Loss of business insurance coverage
- Validates security, availability, and confidentiality
- Builds trust with enterprise healthcare clients
- Proves robust data management practices
- Ensures operational transparency and control
- Aligns with modern data protection laws
- Implement strong security and monitoring controls
- Conduct independent SOC 2 audits annually
- Continuous monitoring of system availability
- Document and enforce data access policies
- Maintain incident response and recovery plans
Our SOC 2 Type II Compliance Partners
ISO 27001
Demonstrates a globally recognized commitment to managing healthcare data risks.
- Increased vulnerability to cyberattacks
- Loss of international market opportunities
- Reputational damage in global healthcare
- Non-compliance with security standards globally
- Barriers to working with multinational clients
- Provides a global framework for information security
- Ensures confidentiality, integrity, and availability of data
- Demonstrates international security compliance
- Essential for global healthcare software vendors
- Reduces risks of operational downtime
- Establish Information Security Management System (ISMS)
- Conduct risk assessments and mitigation plans
- Follow ISO 27001 certification guidelines
- Regular audits and continuous improvements
- Employee awareness and security training programs
Our ISO 27001 Certification Partners
FDA 510(k) Clearance
Proves your software is safe and effective for clinical use under FDA regulations.
- Prohibition from marketing or selling the product
- FDA warnings, recalls, or legal action
- Financial losses and delayed launches
- Severe patient safety risks
- Mandatory product redesign or withdrawal
- Validates the safety and effectiveness of solutions
- Mandatory for SaMD and clinical devices
- Builds trust with hospitals and clinicians
- Enables legal U.S. market clearance
- Prevents patient harm from unsafe tools
- Determine device/software classification
- Conduct clinical risk and safety assessments
- Prepare and submit FDA 510(k) documentation
- Support post-market surveillance and reporting
- Maintain compliance with FDA updates and recalls
Our FDA 510 (k) Compliance Partners